Is Agno3 Nacl Agcl Nano3 an Oxidation Reduction Reaction
Oxidation numbers of the individual element on the product side. Is the reaction NaClaq AgNO3aq NaNO3aq AgCls an oxidation-reduction reaction.

Solved Which Of The Following Is An Oxidation Reduction Chegg Com
Mg cl2 mgcl2 2.

. AgNO3 NaCl AgCl NaNO3 c. On the right side. NaClAgNO3NaNO3AgCl is not a oxidation-reduction reaction because there is no change in oxidation state of any element.
A AgNO3 NaCl AgCl NaNO3 B Cl2 H2O HClO HCl C CuO CO CO2 Cu D NaOH HCl NaCl H2O Which two reactions are redox. IS NaClAgNO3-AgClNaNO3 A REDOX REACTION. Questions asked by students Ask your question and find the answer free.
Ag 1 N 5 O -2. Solve any question of Chemical Reactions and Equations with-. Oxidation involves the LOSS of electrons OIL.
On the left side. There is no change in the oxidation numbers hence it is not an oxidation-reduction reaction. Hno3 naoh nano3 h2o 4.
Asked Jun 30 2017 in Environmental Atmospheric Sciences by Marcia. None of these c. The nitrate ion is unchanged as are the sodium silver and chloride ions.
Which of the following is not oxidation reaction. IS NaClAgNO 3 -AgClNaNO 3 A REDOX REACTION. Oxidation numbers of the individual element on the reactant side.
KOH HNO3 H2O KNO3 b. In this case AgCl is. In this case AgCl is the precipitate.
The equation for the reaction is below. During this reaction the cations and anions of two different compounds switch places forming two entirely different compounds. When solutions of AgNO3 and NaCl react the balanced molecular equation is.
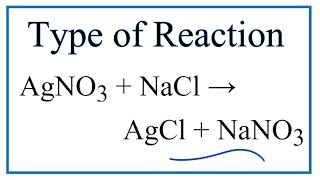
In this video we determine the type of chemical reaction for the equation AgNO3 NaCl AgCl NaNO3 Silver Nitrate and Sodium Chloride. Synthesis reaction Does NaCl react with AgNO3. Which of the following is an oxidation-reduction reaction.
The reaction AgNO3aq NaClaq AgCls NaNO3aq is an reaction. In this set of chemical reactions which reaction is an oxidation-reduction reaction. AgCl is a precipitate and does not cause for hydrolysis reaction.
The products of the reaction are AgCl and NaNO 3. StackrelIHstackrel-IClaqstackrelINa stackrel-IIOstackrelIHaq rarr stackrelINa stackrel-I Claq. Ag 1 Cl -1.
Double displacement reaction is not an oxidation reaction. In the equation for an endothermic reaction the word heat appears. Which one of the following reactions is an oxidation reduction reaction.
AgNO3 NaCl AgCl NaNO3. Both NaCl and AgNO 3 are neutral solutions. AgNO3aq NaClaq AgCls NaNO3aq A 1079 g B 1699 g C 844 g D 0589 g E 589 g.
In a redox reaction oxidation number of. Given the equation 2CO O2 2CO2 how many grams of CO2 form if 340 grams of CO are combined with excess oxygen. In the reaction of silver nitrate with sodium chloride how many grams of silver chloride will be produced from 100.
AgNO3aq NaClaq AgCls NaNO3a. Which of the following is an oxidation reduction reaction. Also NaNO 3 is a neutral compound.
N2 O2 2NO. Nacl agno3 agcl nano3 Answers The answer is one always pick the equation that has at least one single element Correct answer is 1 Explanation. Certified by MeritNation Expert Prafull added an answer on 18713 No Because nothing has been oxidised or reduced.
As an example silver nitrate and sodium chloride react to form sodium nitrate and the insoluble compound silver chloride. C N2 O2 ---- 2NO. On the left side.
Estimate the temperature where Delta G 0 for the following reaction Given. CaCl2 Na2SO4 CaSO4 2NaCl d. G of silver nitrate when it is mixed with an excess of sodium chloride.
Both oxidation and reduction d. Cao h2o ca oh2 3. Is a redox reaction.
Therefore final solution should have a pH value close to 7. AgNO3aq NaClaq AgCls NaNO3aq How much AgCl is produced when 310 g of AgNO3 and 0600 g of NaCl react. In the reaction agno3 aq nacl aq nano3 aq agcl s the reactants a.
A CaCl2 Na2SO4 ----- CaSO4 2NaCl. Which is not an oxidation-reduction reaction. Na 1 Cl -1.
NaCl AgNO 3 AgCl NaNO 3. No Because nothing has been oxidised or reduced. Grades 10 12 Redox Balancing Redox Reactions Practice Worksheet In This Hands On Worksheet Students Will Be R Redox Reactions Reactions Practices Worksheets The loss of electrons results in an increase in charge or oxidation state.
Correct options are A and B This reaction is both precipitation reaction as well as double displacement reaction. Delta H-176 and Delta S -2845 JK NH3 HCl -- NH4Cl. B KOH HNO3 ---- H2O KNO3.
What happens when NaCl is mixed with AgNO3. Since we have two. Consider the equations A B C and D.
Double displacement reactions may be defined as the chemical reactions in which one component each of both the reacting molecules is exchanged to form the products. This reaction is both precipitation reaction as well as double displacement reaction. There has been no exchange of electron This conversation is already closed by Expert Was this answer helpful.
In a double displacement reaction the anions are exchanged and in a precipitation reaction a precipitate is formed in the end. NaCl AgNO3 AgCl NaNO3 NaNO2 HCI - NaCl HNO2 CaSO3 2HCl - CaCl2 H20S02 3CuO 2NH3 - 3Cu 3H2O N2 Use Faradays constant to calculate the charge in Coulombs used during the electrolysis for 000964 mol e F 96500 coulombs per mole. Al2SO43 6KOH 2AlOH3 3K2SO4 e.
A K2SO4 BaCl2 BaSO4 2KCl b CuSO4 BaCl2 BaSO4 CuCl2 c Zn H2SO4 ZnSO4 H2 d PbNO32 2NaCl PbCl2 2NaNO3 e AgNO3 NaCl AgCl NaNO3. Redox reactions involve formal electron transfer and a FORMAL change in oxidation number. So we write the individual reactions with the oxidation number of each constituent species superscripted.
Which type of reaction is NaCl. In a double displacement reaction the anions are exchanged and in a precipitation reaction a precipitate is formed in the end.

Solved Which Is An Oxidation Reduction Reaction A 4na O2 Chegg Com

No comments for "Is Agno3 Nacl Agcl Nano3 an Oxidation Reduction Reaction"
Post a Comment